NeuraLace Medical - About the company
NeuraLace Medical is a seed company based in San Diego (United States), founded in 2016 by Shiv Shukla. It operates as a Non-invasive neuromodulation for chronic neuropathic pain relief and improved patient outcomes. NeuraLace Medical has raised $5.85M in funding from FusionX Ventures. The company has 3003 active competitors, including 937 funded and 706 that have exited. Its top competitors include companies like Jazz Pharmaceuticals, Illumina and Karyopharm.
Company Details
Non-invasive neuromodulation for chronic neuropathic pain relief and improved patient outcomes. They are focused on revolutionizing chronic pain management through its electroceutical platform, offering a non-invasive alternative to traditional methods. Their Axon Therapy mPNS technology uses magnetic peripheral nerve stimulation to recondition the central nervous system, providing long-term relief from post-trauma and post-surgical neuropathic pain. This innovative approach minimizes dependence on opioids and other medications, improving patients' quality of life. The company's technology is FDA-cleared and supported by clinical trial results, demonstrating significant effects in pain reduction. They are dedicated to freeing the world from chronic neuropathic suffering through advanced neuromodulation technology.
- Website
- neuralacemedical.com
- Phone Number
- +1 **********
Key Metrics
Founded Year
2016
Location
San Diego, United States
Stage
Seed
Total Funding
$5.85M in 2 rounds
Latest Funding Round
Investors
Ranked
1098th among 3003 active competitors
Employee Count
22 as on Mar 31, 2026
Legal entities associated with NeuraLace Medical
NeuraLace Medical is associated with 2 legal entities given below:
Legal Entity Name | Date of Incorporation | Revenue | Latest Employee Count | Documents |
|---|---|---|---|---|
NEURALACE MEDICAL INC. CIN: 32090307656 , United States, Active | Jun 13, 2023 | - | - | - |
Neuralace Medical, Inc. CIN: 6131612 , United States, Active | Aug 23, 2016 | - | - | - |
Sign up to download NeuraLace Medical's company profile
NeuraLace Medical's funding and investors
NeuraLace Medical has raised a total funding of $5.85M over 2 rounds. Its first funding round was on Mar 15, 2017. Its latest funding round was a Seed round on Jun 19, 2019 for $*****. 4 investors participated in its latest round. NeuraLace Medical has 8 institutional investors.
Here is the list of recent funding rounds of NeuraLace Medical:
Date of Funding | Funding Amount | Round Name | Post-Money Valuation | Revenue Multiple | Lead Investors | Other Investors |
|---|---|---|---|---|---|---|
Jun 19, 2019 | 1725407 | Seed | 1316020 | 2740382 | 1192360 | 3194878 |
Mar 15, 2017 | 2289035 | Seed | 8537873 | 7319672 |
View details of NeuraLace Medical's funding rounds and investors
NeuraLace Medical's founders and board of directors
Founder? Claim ProfileThe founders of NeuraLace Medical is Shiv Shukla.
Here are the details of NeuraLace Medical's key team members:
- Shiv Shukla: Founder & CEO of NeuraLace Medical and founder of 1 other company.
View details of NeuraLace Medical's Founder profiles and Board Members
NeuraLace Medical's employee count trend
NeuraLace Medical has 22 employees as of Mar 26. Here is NeuraLace Medical's employee count trend over the years: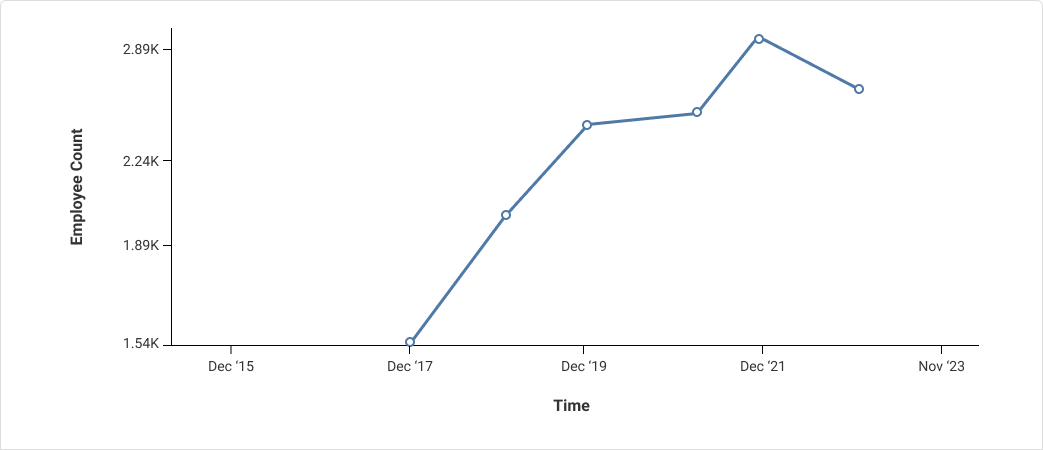

Access Tracxn on any website
Install Tracxn ExtensionOur Google Chrome extension lets you view company details while browsing their websites
NeuraLace Medical's Competitors and alternates
Top competitors of NeuraLace Medical include Jazz Pharmaceuticals, Illumina and Karyopharm. Here is the list of Top 10 competitors of NeuraLace Medical, ranked by Tracxn score:
 Get insights and benchmarks for competitors of 2M+ companies! Sign up today!
Get insights and benchmarks for competitors of 2M+ companies! Sign up today!
Rank | Company Details | Short Description | Total Funding | Investors | Tracxn Score |
|---|---|---|---|---|---|
1st | Jazz Pharmaceuticals 2003, Dublin (Ireland), Public | Developer of novel biopharmaceutical medicines for unmet medical needs | $265M | 81/100 | |
2nd | Illumina 1993, San Diego (United States), Public | Developer of technologies for analysis of genetic variation and function | $28M | 81/100 | |
3rd | Karyopharm 2008, Natick (United States), Public | Developer of small molecule drugs for the treatment of cancer and inflammatory disease | $98.4M | 75/100 | |
4th | Developer of antibody-based drugs to treat autoimmune diseases and cancer | $61.9M | 74/100 | ||
5th | C4 Therapeutics 2016, Cambridge (United States), Public | Developer of small molecule drugs for treating cancer | $223M | 74/100 | |
6th | Syndax 2005, Waltham (United States), Public | Developer of targeted therapies for the treatment of cancer | $198M | 74/100 | |
7th | Alector 2013, San Francisco (United States), Public | Developing immuno-modulatory therapies for dementia and other neurodegenerative disorders | $215M | 74/100 | |
8th | Erasca 2018, San Diego (United States), Public | Developer of small molecule based therapeutics for cancer | $300M | 72/100 | |
9th | Landis+Gyr 1896, Zug (Switzerland), Public | Provider of end-to-end energy management solutions | $1.03B | 72/100 | |
10th | Juno Therapeutics 2013, Seattle (United States), Acquired | CAR T-Cell and T-Cell Receptor-based immunotherapies for the treatment of cancer. | $317M | 71/100 | |
1098th | NeuraLace Medical 2016, San Diego (United States), Seed | Non-invasive neuromodulation for chronic neuropathic pain relief and improved patient outcomes | $5.85M | 40/100 |
Looking for more details on NeuraLace Medical's competitors? Click here to see the top ones
NeuraLace Medical's Investments and acquisitions
NeuraLace Medical has made no investments or acquisitions yet.
Reports related to NeuraLace Medical
Here is the latest report on NeuraLace Medical's sector:
News related to NeuraLace Medical
•
Neuralace's Axon Therapy Gets FDA Nod for Painful Diabetic NeuropathyMedical Product Outsourcing•Jan 11, 2024•NeuraLace Medical
•
Neuralace Medical Announces Clinical Milestones Including Presentation of the Safety and Efficacy of Axon Therapy (SEAT) Study at the American Society of Pain and Neuroscience Conference (ASPN), While Completing Enrollment in the Axon Therapy Painful Diabetic Neuropathy Study (AT-PDN) and Launching a Pilot Study Focused on the Treatment of Chemotherapy Induced Peripheral Neuropathy (CIPN)Tullahoma News•May 30, 2023•NeuraLace Medical
•
•
•
•
Neuralace Medical Advises the AMA Issues Unique Category III CPT Code for Transcutaneous Magnetic Nerve StimulationMedical Device News•Jun 29, 2022•NeuraLace Medical
•
NeuraLace Medical Announces Appointment of Keith Warner as Chief Executive OfficerPR Newswire•Mar 26, 2022•NeuraLace Medical,
•
NeuraLace ties up FDA clearance for chronic pain-relieving nerve stimulation deviceFierce Biotech•Jun 21, 2021•NeuraLace Medical, FDA
•
•
NeuraLace Medical Closes $3 Million Seed RoundSan Diego Business Journal•Jun 16, 2019•NeuraLace Medical
Are you a Founder ?
FAQs about NeuraLace Medical
Explore our recently published companies
- Saga - London based, 2017 founded, Series A company
- Sitel - Gurugram based, 1985 founded, Acquired company
- Zapi - Singapore based, 2025 founded, Unfunded company
- CorpChamber - Jakarta based, 2023 founded, Unfunded company
- Oxynum - Paris based, 2012 founded, Unfunded company
- WAVEE - Tokyo based, 2022 founded, Unfunded company
